Clean Dewars and Glass Houses

Donate
At the International IVF Initiative, we are committed to providing free access to our educational sessions, webinars, and resources for professionals and individuals passionate about advancing reproductive medicine. We believe that cost should never be a barrier to knowledge and collaboration. By contributing, you’re ensuring that valuable educational resources, expert insights, and collaborative opportunities remain open to all without financial barriers. Together, we can continue to foster a global community dedicated to innovation and excellence in the field of IVF.
Your Donation
Thank you!
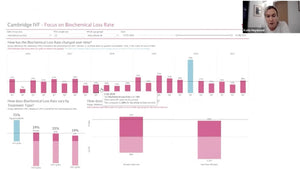
Clean Dewars and Glass Houses
Tuesday 27th April, 2021. 3PM EST/ 8PM GMT / 9PM CET
DR. MARTINE NIJS
 Martine Nijs obtained her Master in Zoology at the Free University of Brussels, Belgium. After obtaining her PhD in Medical Sciences at the Free University of Brussels in Belgium, Martine has been working as a senior clinical embryologist and later as an IVF lab director and Scientific Director.
Martine Nijs obtained her Master in Zoology at the Free University of Brussels, Belgium. After obtaining her PhD in Medical Sciences at the Free University of Brussels in Belgium, Martine has been working as a senior clinical embryologist and later as an IVF lab director and Scientific Director. Dr. Nijs is an ESHRE accredited senior clinical embryologist, and past Scientific, IVF and Andrology Lab Director in Nij Geertgen, Netherlands, Lab Director at the Genk Institute for Fertility Technology, Belgium and Senior Embryologist at the Schoysman Infertility Management Team in Belgium.
She is the author of numerous peer-reviewed publications and book chapters and has presented hundreds of lectures as an invited speaker at meetings worldwide. She has been actively involved in organizing international conferences, workshops, and one-to-one practical training. Martine is an ad hoc board member of 10 international journals and an active member of 8 professional societies.
Martine is currently the Global Training Manager at CooperSurgical Fertility Solutions.
Bibliography:
Alikani M, Parmegiani L. Human Reproductive Cell Cryopreservation, Storage, Handling, and Transport: Risks and Risk Management. Semin Reprod Med. 2018 Sep;36(5):265-272. doi: 10.1055/s-0038-1676849. Epub 2019 Apr 4. PMID: 30947342.
Bielanski, S. Nadin-Davis, T. Sapp, and C. Lutze-Wallace, Viral contamination of embryos cryopreserved in liquid nitro gen,Cryobiology, vol. 40, no. 2, pp. 110–116, 2000.
Bielanski A. Non-transmission of bacterial and viral microbes to embryos and semen stored in the vapour phase of liquid nitrogen in dry shippers. Cryobiology. 2005 Apr;50(2):206-10. doi: 10.1016/j.cryobiol.2004.12.004. PMID: 15843010.
Bielanski A, Vajta G. Risk of contamination of germplasm during cryopreservation and cryobanking in IVF units. Hum Reprod. 2009 Oct;24(10):2457-67. doi: 10.1093/humrep/dep117. Epub 2009 Jun 26. PMID: 19561041.
Bielanski A. Biosafety in embryos and semen cryopreservation, storage, management and transport. Adv Exp Med Biol. 2014;753:429-65. doi: 10.1007/978-1-4939-0820-2_17. PMID: 25091919.2009
Cobo, J. Bellver, M. J. De Los Santos, and J. Remoh´ı, “Viral screening of spent culture media and liquid nitrogen samples of oocytes and embryos from hepatitis B, hepatitis C, and human immunodeficiency virus chronically infected women undergoing in vitro fertilization cycles,” Fertility and Sterility, vol. 97, no. 1, pp. 74–78, 2012.
Clarke GN. Sperm cryopreservation: is there a significant risk of cross-contamination? Hum Reprod 1999;14:2941–2943.
College of American Pathologists Accreditation Checklist, College of American Pathologists 2019. Available at: https://www.cap.org/memberresources/ practice-management. Accessed July 1, 2020.
Fountain D, Ralston M, Higgins N, Gorlin JB, Wheeler C, Antin JH, Churchill WH. Liquid nitrogen freezers: a potential source of microbial contamination of hematopoietic stem cell components. Transfusion 1997;37:585–591.
Fountain, D.; Ralston, M.; Higgins, N.; Gorlin, J. B.; Uhl, L.; Wheeler, C.; Antin, J. H.; Churchill, W. H. and Benjamin, R. J. 1997. Liquid nitrogen freezers: A potential source of microbial contamination of hematopoietic stem cell components. Transfusion 37:585-591
Go KJ. A subspecialty of the assisted reproductive technologies: cryogenic inventory maintenance. J Assist Reprod Genet. 2019 Jan;36(1):15-17. doi: 10.1007/s10815-018-1378-z. Epub 2018 Dec 15. PMID: 30554394; PMCID: PMC6338608
Isachenko V, Isachenko E, Montag M, Zaeva V, Krivokharchenko I, Nawroth F, Dessole S, Katkov II, van der Ven H. Clean technique for cryoprotectant-free vitrification of human spermatozoa. Reprod Biomed Online. 2005 Mar;10(3):350-4. doi: 10.1016/s1472-6483(10)61795-6. PMID: 15820041.
Letur-Könirsch H, Collin G, Sifer C, Devaux A, Kuttenn F, Madelenat P, Brun-Vezinet F, Feldmann G, Benifla JL. Safety of cryopreservation straws for human gametes or embryos: a study with human immunodeficiency virus-1 under cryopreservation conditions. Hum Reprod 2003;18:140–144.
Loskutoff NM, Huyser C, Singh R, Morfeld KA, Walker DL, Thornhill AR, Smith M, Morris L, Webber L. Removing HIV-1 and hepatitis C virus from semen: an improved method for washing sperm using a trypsinized silica particle suspension and a novel tube insert. Fertil Steril 2005;84:1001–1010.
Morris, G. J. 2005. The origin, ultra structure, and microbiology of the sediment accumulating in liquid nitrogen storage vessels. Cryobiology 50:231-238.
Mortimer D. Current and future concepts and practices in human sperm cryobanking. Reprod. Biomed. Online 2004;9:134–151.
Pomeroy KO, Harris S, Conaghan J, et al. Storage of cryopreserved reproductive tissues: evidence that cross-contamination of infectious agents is a negligible risk. Fertil Steril 2010;94(04): 1181–1188
Practice Committees of the American Society for Reproductive Medicine, Society for Reproductive Biologists and Technologists, and Society for Assisted Reproductive Technology. Cryostorage of reproductive tissues in the in vitro fertilization laboratory: a committee opinion. Fertil Steril. 2020 Sep;114(3):486-491. doi: 10.1016/j.fertnstert.2020.06.019. Epub 2020 Aug 7. PMID: 32778330.
Rienzi L, Gracia C, Maggiulli R, LaBarbera AR, Kaser DJ, Ubaldi FM, Vanderpoel S, Racowsky C. Oocyte, embryo and blastocyst cryopreservation in ART: systematic review and meta-analysis comparing slow-freezing versus vitrification to produce evidence for the development of global guidance. Hum Reprod Update. 2017 Mar 1;23(2):139-155. doi: 10.1093/humupd/dmw038. PMID: 27827818; PMCID: PMC5850862.
Russell PH, Lyaruu VH, Millar JD, Curry MR, Watson PF. The potential transmission of infectious agents by semen packaging during storage for artificial insemination. Anim Reprod Sci 1997;47:337–342.
Schiewe MC, Freeman M, Whitney JB, VerMilyea MD, Jones A, Aguirre M, Leisinger C, Adaniya G, Synder N, Chilton R, Behnke EJ. Comprehensive assessment of cryogenic storage risk and quality management concerns: best practice guidelines for ART labs. J Assist Reprod Genet. 2019 Jan;36(1):5-14. doi: 10.1007/s10815-018-1310-6. Epub 2018 Sep 19. PMID: 30232640; PMCID: PMC6338590.
Selman H,Mariani M, Barnocchi N, et al. Examination of bacterial contamination at the time of embryo transfer, and its impact on the IVF/pregnancy outcome. J Assist Reprod Genet 2007;24(09): 395–399