Making Time for Compliance

Donate
At the International IVF Initiative, we are committed to providing free access to our educational sessions, webinars, and resources for professionals and individuals passionate about advancing reproductive medicine. We believe that cost should never be a barrier to knowledge and collaboration. By contributing, you’re ensuring that valuable educational resources, expert insights, and collaborative opportunities remain open to all without financial barriers. Together, we can continue to foster a global community dedicated to innovation and excellence in the field of IVF.
Your Donation
Thank you!
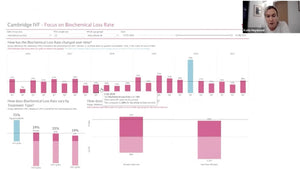
Session 71: Making Time for Compliance
Tuesday 13th July, 2021. 3PM EST/ 8PM GMT / 9PM CET
A session with a mix of short talks and lively discussions about staffing levels, workload and the changing role of the embryologist.
CASSIE MILLER
 Cassie Miller, Ovation® Fertility’s director of risk management, helps Ovation’s IVF labs maintain regulatory compliance while creating opportunities for next-level information and data sharing to drive performance improvement and reduce risk across the organization.
Cassie Miller, Ovation® Fertility’s director of risk management, helps Ovation’s IVF labs maintain regulatory compliance while creating opportunities for next-level information and data sharing to drive performance improvement and reduce risk across the organization.
Miller’s primary focus is ensuring that Ovation’s employees have what they need to work safely, that embryos are protected, and that patient identification and privacy are maintained. By gathering, analyzing and sharing organization wide quality management data, she proactively identifies risks and implements appropriate preventative actions to minimize, mitigate or eliminate issues before they occur.
Miller joined Ovation in July 2019 as compliance manager, ensuring that each of Ovation’s laboratories were in compliance with all rules, regulations and laws that relate to the fertility care field. In this role, she standardized Ovation’s regulatory response and helped Ovation labs address ever-changing standards for privacy and safety, helping to ensure delivery of high-quality patient care.
Before joining Ovation, Miller served as Midwest territory manager for Irvine Scientific. She also has more than 20 years of hands-on clinical laboratory experience, including three years as an embryologist, andrologist, endocrinology specialist, and interim supervisor for Bennett Fertility Institute in Oklahoma City.
Earlier in her career, she spent more than a decade as a laboratory supervisor, lead medical technologist and blood bank supervisor for OU Medicine, Inc., in Edmond, Oklahoma. Prior to that, she worked for nearly nine years as a lead medical technologist for AllianceHealth Deaconess in Oklahoma City.
Certified as a medical technologist by the American Society for Clinical Pathology (ASCP), Miller earned a Bachelor of Science degree in clinical laboratory science and medical technology from the University of Central Oklahoma.