Management of IVF; Electronic Applications to the Rescue

Donate
At the International IVF Initiative, we are committed to providing free access to our educational sessions, webinars, and resources for professionals and individuals passionate about advancing reproductive medicine. We believe that cost should never be a barrier to knowledge and collaboration. By contributing, you’re ensuring that valuable educational resources, expert insights, and collaborative opportunities remain open to all without financial barriers. Together, we can continue to foster a global community dedicated to innovation and excellence in the field of IVF.
Your Donation
Thank you!
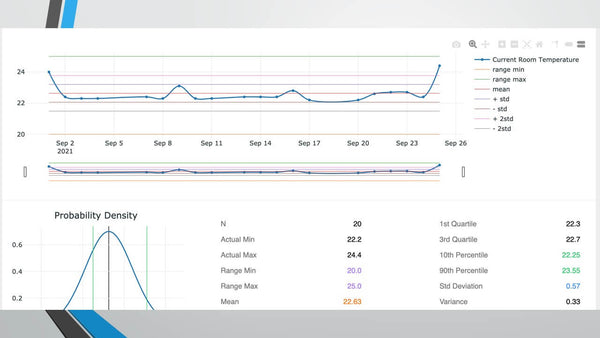
Management of IVF; Electronic Applications to the Rescue
Tuesday 28th September, 2021. 3PM EST/ 8PM GMT / 9PM CET
MS. UNIKA ALEXANDRA

She is a Clinical Laboratory Scientist licensed by American Society of Clinical Pathology with 10 years of experience in reproductive medicine.
She previously worked with California Cryobank as a Branch Manager, Laboratory Operations Manager and Tissue Bank Director but currently working with Northwell Health Fertility as the Clinical Laboratory Operations Manager.